HOLLOW CATHODE LAMP ELAPSED TIME RECORDING SYSTEM - diagram, schematic. While elements having various melting points are used, more volatile element is lost first resulting within gradual weakening of its spectrum and degeneration within one element lamp. HOLLOW CATHODE LAMP ELAPSED TIME RECORDING SYSTEM - diagram, schematic, and image 05. In that case, cathode is made up from alloys of metals having same melting point such as Ca-Mg, Ag-Au, Cu-Fe, Zn-Cd, etc. Therefore, these days multielement cathode lamps are more in use for routine determinations, by their performance is not extremely reliable.  
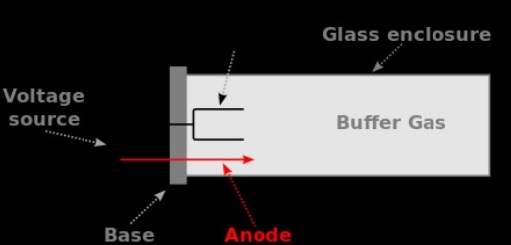
As of now, HCL for over 60 elements are available. Intensity of resonance lines from an HCL rises with increasing current. The emission spectrum of the cathode material involves a number of intense, sharp lines because of transitions among excited states and the ground state, frequently known as resonance lines. These metal atoms are then excited through collisions along with electrons and ions therefore emitting features emission lines. Collision energy is enough to cause some atoms of the cathode to be transformed within gaseous atoms in a procedure known as sputtering. Other line sources such as electrode-less discharge lamps are frequently used for determination of nonmetals. Figure 10 illustrates the basic construction details of these lamps. For that the gaseous cations are accelerated towards the cathode. The hollow cathode lamp: The most important aspect of AAS instrumentation is the light source, the most common of which is the hollow cathode lamp (HCL). While a voltage of ~300 V corresponding to 5-50 m A current is applied among the two electrodes, a low pressure glow discharge confined to the within of the cathode material is generates.įigure: Schematic diagram of hollow cathode lamp illustrating different componentsīasic function of the gas within the tube is to bombard the cathode and vapourise the atoms from the cathode surface. The schematic diagram of a hollow-cathode lamp is displays in Figure. The cathode is fabricated from the analyte element and the lamp is filled along with an inert gas such as argon or neon under vacuum (100-200 Pa). It consists of a sealed cylindrical glass tube along with a quartz window at one end and a hollowed cylindrical cathode together within an anode wire made of tungsten. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
